EIQ deploys Ai detection tech into New York’s no.1 cardiology hospital
Our AI heart disease detection Investment EchoIQ (ASX: EIQ), just rolled out its AI-based heart disease detection software into another leading US health system in New York…
EIQ deployed its tech into the Mount Sinai Health System, which comprises seven hospitals, over 400 outpatient practices and +3,760 beds.
That’s the same hospital is actually ranked no.1 in New York and no.2 in the USA for Cardiology, Heart & Vascular Surgery.
EIQ is now 18 months into the rollout of the AS tech, which was cleared by the FDA back in October 2024. (source)
The reason today’s news matters is because:
- It means the Aortic Stenosis tech is in more hospitals, being used more in clinics - which ultimately helps with EIQ’s case for getting higher category reimbursement codes (more on this in a second)
- It means EIQ is building a distribution network that it can utilise for its Heart Failure (HF) tech, which is currently going through the FDA clearance process…
That second reason is the one that we think is the major catalyst for EIQ in the short-term.
EIQ’s HF tech already has established reimbursement code pathways - so it can rollout instantly piggybacking on those existing codes.
Basically, IF the Heart Failure tech is cleared by the FDA - EIQ SHOULD be able to turn it into revenues a lot quicker than its Aortic Stenosis tech - which will need time to hit higher reimbursement code categories.
Because EIQ’s is a software product, it is very much “plug and play”.
Any hospital networks that are already using EIQ for Aortic Stenosis, can easily set up for Heart Failure as well... if/once the FDA clearance comes in.
Right now, EIQ is at the final stage of obtaining FDA approval for its HF tech, it has run the studies and submitted the application back in late December:
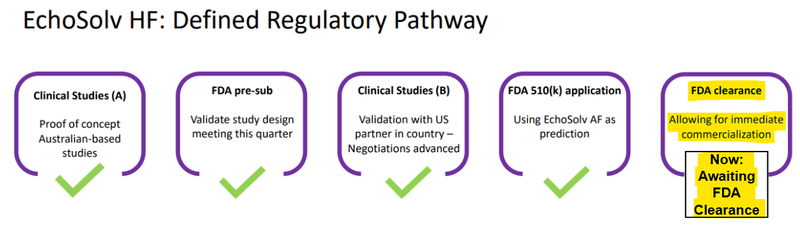
(source)
This has a standard review time of around 90 days, which has now passed:
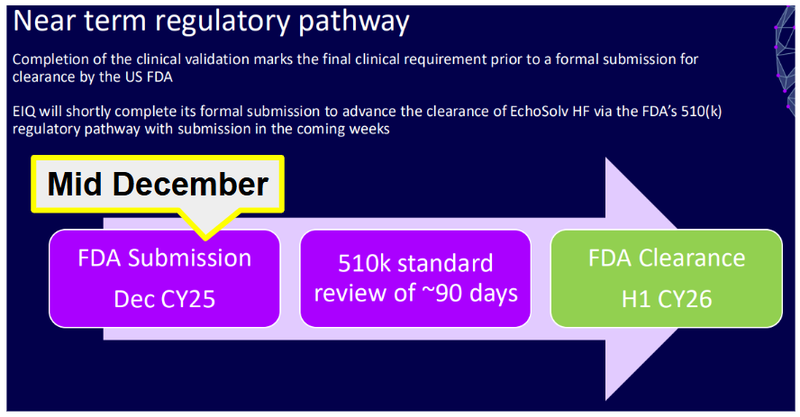
(source)
EIQ previously provided guidance that it was expecting a decision during H1 this year, so this decision could land any day now that there are only 2 months left in the half.
What are reimbursement codes and why they matter
First, what is a reimbursement code?
A reimbursement code is a specific administrative code that allows hospitals and doctors to bill insurance companies (such as Medicare in the US).
So should it get approval, this will allow doctors to be more likely to use EIQ’s technology because they will be able to bill through the insurance company rather than the patient footing all of the cost.
Here is where EIQ’s two technologies sit from a reimbursement perspective:
1. Aortic Stenosis detection tech
At the moment EIQ’s is able to bill for use of its AS tech using a "Miscellaneous” reimbursement code (93799).
“Miscellaneous" codes, requires more documentation/admin work and as a result there is less certainty of reimbursement.
For the Aortic Stenosis tech we want to see EIQ go from Misc code to category III and then eventually category I (the holy grail for reimbursement codes):
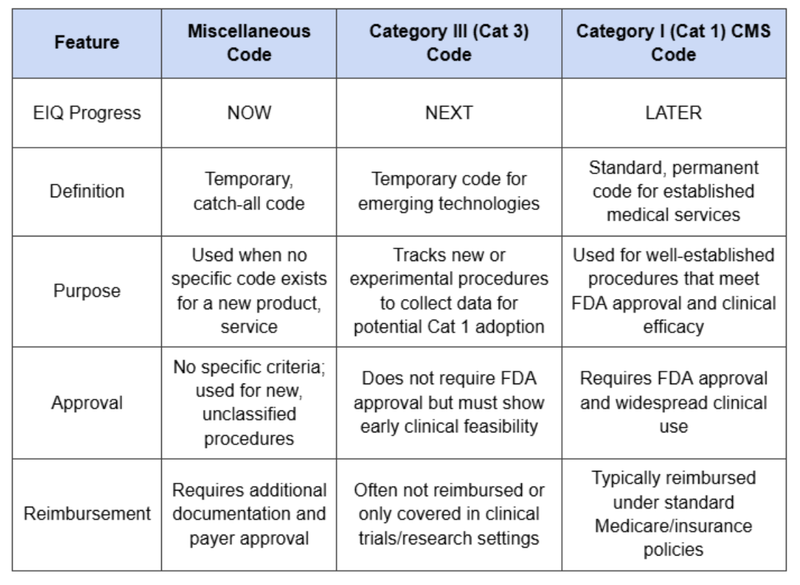
It was unsuccessful last year in its application, however is applying again this year as there is now a new AI specific framework being developed by the AMA which it is following up. (we covered that here)
This tech can (and is) still being used, it is simply easier for doctors/hospitals to charge for the usage if it has its own specific reimbursement code.
2. Heart Failure detection tech
For this technology, there already exists a reimbursement code thanks to a company called Ultromics.

(source - our prior deep dive article)
We have written about the Ultromics comparison before here.
Of course, this doesn’t really matter for EIQ as of right now, given the first hurdle is FDA clearance.
So, IF EIQ can’t get its Heart Failure tech cleared by the FDA, the reimbursement categories available to it won't matter.
Recent results prior to the FDA application were stronger than Ultromics, plus the data came from a dataset of ~13x more patients. (source)(source)
So this may be a reason why the share price has been on a good run this year (up around 300%) with the market likely expecting a positive outcome from this application on the back of the results:
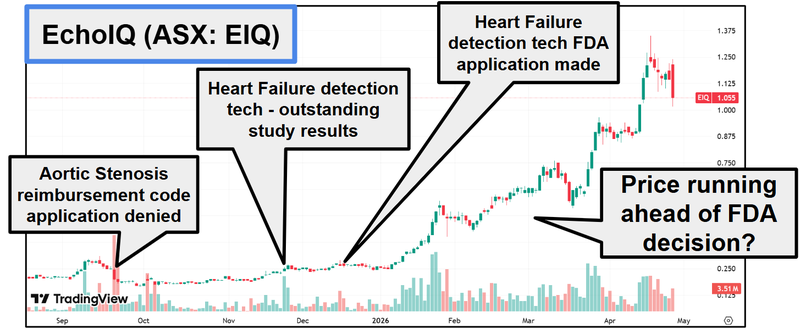
(source)
What’s next for EIQ?
🔄 Heart failure FDA approvals
Next is all about FDA clearance for EIQ’s heart failure detection tech.
As mentioned above, we are expecting to hear back from the FDA inside H1-2026… possibly as early as this month, there are only ~2 months remaining and the average time for a decision (90 days) has now passed.
🔄 Commercialisation updates for Aortic Stenosis AI tech
The key metric we will be tracking in the short term is how many integrations EIQ can secure for its Aortic Stenosis tech.
In the short term we want to see more distribution deals - either through strategic partnerships or reseller deals.
Today we got an update related to this with Mount Sinai Health System (in addition to the Mayo Clinic prior) to include its heart failure detection software, so should FDA clearance come through on this, it can start to be commercialised immediately.
🔄 Australia and NZ pilot program
EIQ has previously mentioned that this program is being run with a ”leading global structural heart innovation company”.
We want to see some more news on this front because we think it could help advance EIQ’s licensing revenue pathway and be a “proof of concept” study that EIQ can take into the US.
🔄 Partnership with European reseller to broaden market exposure
We want to see EIQ expand into new markets like Europe, in a previous webinar EIQ said the company was pursuing this opportunity and last week the company updated that there was progress on this with negotiations underway.
This also includes CE Mark and TGA applications so that EIQ can sell into Europe and Australia.





